Once superhydrophobic surfaces have been manufactured, we can then think about what we can do with the surfaces, both to further understand the science behind hydrophobicity and to think about potential applications.
Self-cleaning Surfaces

Muddying a superhydrophobic surface is far from easy, as we can see by dipping a surface that has been given a superhydrophobic coating into a box of muddy water.
Droplet Impact and
Rebound

When a water droplet impacts on a solid surface, we expect the drop to splash. However, when impacting on a ‘bed of nails‘, droplets completely rebound without any splashing.
Evaporating
Droplets

When a water droplet rests on a microscopic ‘bed of nails’ surface, it will initially form a ball with a high contact angle. However, as the droplet evaporates, the curvature becomes greater, which leads to a greater pressure inside the droplet. This leads to a sudden transition to a state in which the water penetrates between the ‘nails’.
Directing Droplet Motion

If the roughness changes across a surface, the water-repellency will also change. This leads to interesting motion of droplets on the surface.
Super-vortex
Skating

A jet of water directed onto a funnel with a superhydrophobic coating goes round many times more than one without a coating.
Sensing Changes

While the above example used variation in surface roughness to control superhydrophobicity, hydrophobicity can also be controlled by altering external factors such as temperature. A water droplet can be switched from resting on a foam surface to being absorbed into the foam by heating it up. This could potentially be used as an indicator that the temperature had exceeded a critical level at some time in the past, which is of relevance in food storage for example.
Role of Air Layers
When looking up at the surface of a swimming pool from within the water, we see a silver sheen. Some aquatic animals can also be seen to have a silvery sheen in water, indicating that there is air present. This air layer is known as a plastron. Superhydrophobic man-made objects can also have air layers present when immersed in water, again indicated by a silvery sheen. In the following examples, we will look at the effect of these air layers around objects.
Breathing Underwater

Some aquatic animals extract oxygen from water using a layer of air on their body to create a gas-exchange membrane from the water-air interface. Immersed superhydrophobic surfaces naturally have a plastron, leading to the intriguing question of whether humans could ever be able to breathe underwater using a plastron.
Plastron Drag Reduction
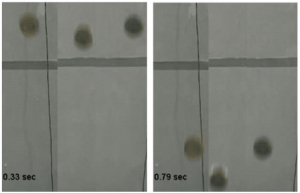
Plastrons aren’t only of relevance for oxygen extraction. Acrylic spheres coated in hydrophobic paint (centre) retain a layer of air around them which results in them falling faster than uncoated spheres (left) and coated spheres treated with ethanol to prevent plastron formation (right).
Flow Through
Pipes

Plastrons can also reduce the friction for water flowing through a pipe. When a flow splits between two pipes, more water will go through the superhydrophobic pipe due to its lower resistance to flow. This is similar to the friction reduction of a hovercraft but without the need for active production of a layer of air.