Liquid Lenses on SLIPS

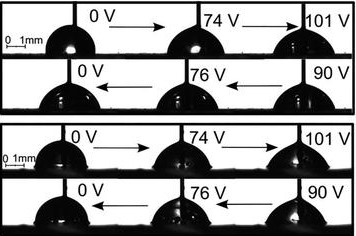
Electric fields can been used to force a droplet to spread on a surface via an applied voltage using electrowetting or dielectrowetting. Electrowetting manipulates the ions in a conducting liquid droplet at the solid-liquid interface and stores capacitive energy by the polarization of a solid dielectric layer. Dielectrowetting manipulates dipoles in a dielectric liquid droplet at the solid-liquid interface to store capacitive energy by polarizing a liquid dielectric layer. We have shown that dielectrowetting on a SLIP surface allows a droplet to be smoothly and reversibly spread into either a film or a droplet with different curvature by application of a suitable voltage. This can be seen in the figure above. Because the spreading is reversible, the droplet can be used as a variable-focus liquid lens.
Use of a SLIP surface is essential for achieving complete and reversible spreading in air. Dielectrowetting can be used to induce complete spreading to a film state, but when used in air, removing the voltage doesn’t result in recovery of the same shape as the original droplet. This is illustrated in the figure below, which shows spreading of a droplet of glycerol on a hydrophobic surface. We can see that the final state of the droplet is not as tall as the original droplet.

Comparson between electrowetting and dielectrowetting
It is interesting to compare the behaviour under dielectrowetting with that under electrowetting. With electrowetting, a glycerol droplet will not completely spread into a film on either a hydrophobic or a SLIP surface. This can be seen in the figure below. On both surfaces, the contact angle of the droplet decreases to a minimum of around 70° to 75° as the voltage is increased, but the angle will not reduce further.
Publication
Electric field induced reversible spreading of droplets into films on lubricant impregnated surfacesZ. Brabcova, G. McHale, G.G. Wells, C.V. Brown and M.I. Newton, Applied Physics Letters 110 (2017) 121603.
